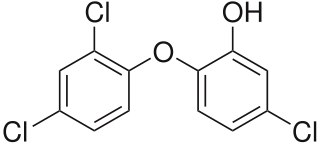
The FDA has finally come out with some strong words regarding Triclosan, the active ingredient of almost all consumer products labeled as “antibacterial.” Triclosan has been the target one of my personal crusades ever since I began studying the science of cleaning chemistry. Originally developed as a pre-surgery scrub for medical professionals, it is an effective anti-microbial agent and fungicide that doesn’t have the side effect of causing dry skin like alcohol- or peroxide-based hand sanitizers. It kills germs by interfering with enzymes necessary for fatty-acid synthesis.
The problem with Triclosan (and other antibiotics like Penicillin and Methicillin) is that it is also a mutagen- it actually causes germs to mutate and develop new characteristics. And every once in a while these mutations create a “Super-Bug,” a germ strain that is resistant to anti-biotics. For example, the deadly MRSA (reaistant staph), is such a super-bug.
Triclosan is most often found in anti-bacterial soaps, toothpaste, and shampoo. It is also present in many municipal water sources, odor-resistant athletic clothes and equipment, and even children’s toys. It interferes with the human body’s endocrine system (it kills our cells the same way it kills bacteria). Because it has snuck its way into nearly every corner of the consumer goods market, it is regulated by the FDA, the EPA, and the Consumer Product Safety Commission.
Luckily, the FDA has finally caught on to the problem. The Washington Post has a great article:
…the FDA said that recent scientific studies raise questions about whether triclosan disrupts the body’s endocrine system and whether it helps to create bacteria that are resistant to antibiotics. An advisory panel to the FDA said in 2005 that there was no evidence the antibacterial soaps work better than regular soap and water. (emphasis added.)
